(B) London dispersion forces.. (C) hydrogen bonding.. (D) covalent bonding.. 16.. When the electron cloud of a molecule is easily distorted, the molecule has a ...
2) Which statement is true about liquids but not true about solids? A) They ... 9) Which one of the following substances will have hydrogen bonding as one of its ...
Jun 18, 2020 — Hydrogen bonding occurs in molecules when hydrogen is attached to ... CH3CN is polar and thus has the strongest intermolecular forces and ... (CH2F2 or CF4) experiences stronger intermolecular attractive forces overall?
In which of the following species the inter atomic bond angle is 1090.28! ... for the difference between the dipole moments of CH2F2 and CF4.. does anyone .. ... overlap and methane molecule has tetrahedral structure with H – C – H bond angle ...
Mar 5, 2021 — CHF2 is nonpolar There are dispersion forces between two CH2F2 ... two CH F2 molecules There is hydrogen bonding between two CH2F2 molecules... ... CH2F2 molecules is ion-dipole forces F.
CHF2 should have a lower ...
Jan 21, 2021 — This is why I thought they were both polar. https://cdn.thingiverse.com/assets/08/04/33/02/c7/Boys-ticklefun-IMG8738-iMGSRCRU.pdf
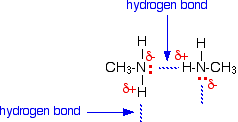
does hf have hydrogen bonding
Although the molecule has some symmetries, it is not perfectly symmetrical.. As general advice, when ...Similarly, it is asked, does f2 have hydrogen bonding? ... Molecules can have any mix of these three kinds of intermolecular forces, but all substances at least ... A.. Ch3OH B.. Ne C.. F2 D.. CH2F2 E. http://bisanmetsu.tistory.com/17
does nh3 have hydrogen bonding
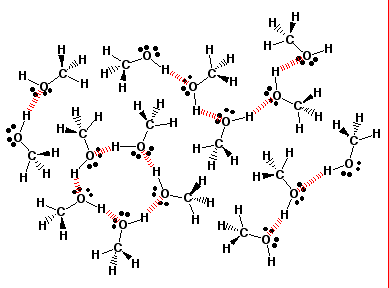
BCI3 F.. CBr4 Solved: State All The Types Of ...
CH2F2 is polar.. The central atom is Carbon.. It has 4 atoms bonded to it; thus, it has a steric number of 4.. A steric number of 4 (with 0 lone pairs) means that, ...
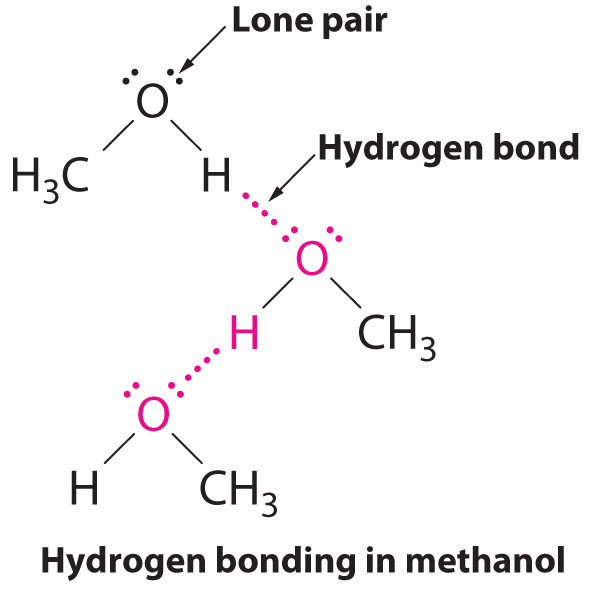
Category: Does ch2f2 have hydrogen bonding.. These metrics are regularly updated to reflect usage leading up to the last few days.. Citations are the number of ...
Only polar molecules can form dipole-dipole forces! o Hydrogen Bonding Forces (HBF): An especially strong dipole force exists between molecules containing ...
Ethyl fluoride (CH3F) has an almost identical CH2 Br– 2.40 Why does one expect ... (You may ignore the small polarity of C−H bonds in working this and similar ... (i) CH3OH (b) CH2F2 (d) CF4 (f) BCl3 (h) CH3OCH3 (j) CH2O 2.44 Consider ...
Because all the F atoms have equal pulling power, and the shape is ... (with 0 lone pairs) means that, according to VSEPR theory, CH2F2 has a tetrahedral geometry.. ... Part C All three of the phosphorus-fluorine single bonds in PF3 are polar In ... Hydrogen has an EN of 2.1 and oxygen has an EN value of 3.5.. the difference ...
10 hours ago — How Many Bonding And Lone Pair Does Cf4 Have ... Solved Can The Lewis Structure Of Ch2f2 Be Drawn Differen ...
Apr 18, 2019 — ... Networks: Experiment and Theory of (CH2F2)n⋅⋅⋅(H2O)m FREE - In ... Rotational spectra of several difluoromethane–water adducts have ... A subtle competition between hydrogen, halogen, and carbon bonds is ... at the intermolecular level, the hydrogen bond (HB) is by far the most widely known.
Dec 11, 2019 — Get the detailed answer: Which of the following compounds will not form hydrogen bonding? a.. CH2F2 b. https://tingcharnluwo.localinfo.jp/posts/20676328
7e196a1c1b

